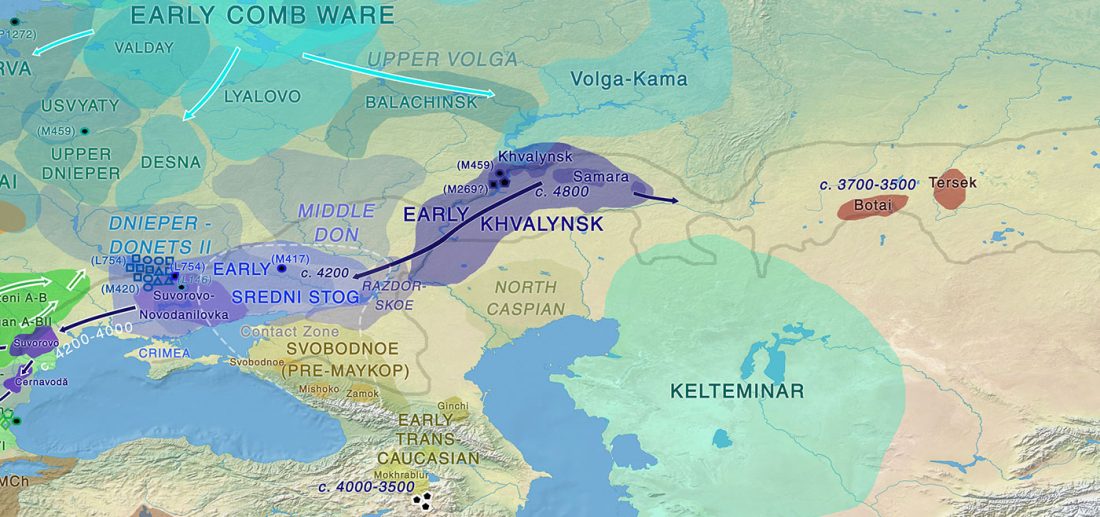
This post is part of a draft on palaeolinguistics and the Proto-Uralic homeland. See below for the color code of protoforms.
7. Animal Husbandry
7.1. Milk
PFU (Saa., Fi., Md., Ma., P, Kh., Ms. Hu.) *waji ‘butter/grease’ (UEW Nº 1156).
NOTE. Aikio (2019: 48) contra Häkkinen (2009: 28-30) considers an original meaning ‘grease’ more likely than ‘butter’. Indeed, a more general meaning is always the most conservative approach. In fact, it was possibly borrowed from Smy. into PTurk. *bań ‘fat, grease; edible oil, butter; lubricant’ (Czentnár 2015: 9-17). What is striking about Aikio’s comments … Read the rest “Proto-Uralic Homeland (IV): Animal Husbandry”